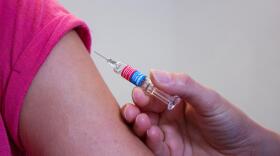
Mass vaccinations and clinical trials are converging and challenging researchers.
Dr. Anna Lok is the assistant dean for clinical research at Michigan Medicine. The health system is part of two COVID-19 vaccine trials: AstraZeneca's, and Johnson & Johnson's.
Lok says the two trials recruited about 400 patients. Then the Pfizer vaccine became available. Then came the phone calls and emails.
"For a couple of weeks, we were working night and day, over the weekend, dealing with all these people saying that, ‘I have been given an appointment tomorrow, I need the unblinding tonight.’"
She says more than a third of participants have asked to be unblinded -- that is, learn whether they received the vaccine or a placebo. Many were health care workers.
But, Lok also noted that Michigan Medicine is just one of many sites taking part in the international trials.